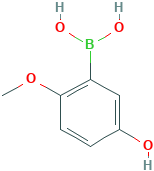
Boronic acid, B-(5-hydroxy-2-methoxyphenyl)-
5-Hydroxy-2-methoxyphenylboronic acid
CAS: 1072952-43-2
Molecular Formula: C7H9BO4
Boronic acid, B-(5-hydroxy-2-methoxyphenyl)- - Names and Identifiers
Boronic acid, B-(5-hydroxy-2-methoxyphenyl)- - Physico-chemical Properties
| Molecular Formula | C7H9BO4 |
| Molar Mass | 167.96 |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Sensitive | Irritant |
| MDL | MFCD07368831 |
Boronic acid, B-(5-hydroxy-2-methoxyphenyl)- - Introduction
It is an organic boron compound with the chemical formula C7H9BO4.
Nature:
a white to light yellow crystalline solid. It is stable at room temperature and soluble in some organic solvents such as dimethyl sulfoxide and dichloromethane. Its solubility in water is low.
Use:
Ti acid is widely used in organic synthesis, especially the riveting reaction. It is an important boronic acid activation reagent, which can react with hydrogen, oxygen or halogen atoms in organic compounds to form borate esters (boronic ester), thus participating in the reaction of C- C bond formation, such as Suzuki coupling reaction.
Preparation Method:
Cr acid can be synthesized by acid-catalyzed reaction of phenylboronic acid and ortho-or meta-hydroxylation. In the laboratory, commonly used benzene boron chloride as the starting material, and ortho or meta hydroxyl compounds such as catechol reaction, hydrolysis treatment to obtain the target product.
Safety Information:
The acid is a chemical and should follow the appropriate laboratory procedures. It may cause eye and skin irritation, so care should be taken to avoid contact when using it. Avoid inhaling dust and vapors and ventilate properly. Storage should be placed in a dry, cool place, away from fire and oxidant.
Nature:
a white to light yellow crystalline solid. It is stable at room temperature and soluble in some organic solvents such as dimethyl sulfoxide and dichloromethane. Its solubility in water is low.
Use:
Ti acid is widely used in organic synthesis, especially the riveting reaction. It is an important boronic acid activation reagent, which can react with hydrogen, oxygen or halogen atoms in organic compounds to form borate esters (boronic ester), thus participating in the reaction of C- C bond formation, such as Suzuki coupling reaction.
Preparation Method:
Cr acid can be synthesized by acid-catalyzed reaction of phenylboronic acid and ortho-or meta-hydroxylation. In the laboratory, commonly used benzene boron chloride as the starting material, and ortho or meta hydroxyl compounds such as catechol reaction, hydrolysis treatment to obtain the target product.
Safety Information:
The acid is a chemical and should follow the appropriate laboratory procedures. It may cause eye and skin irritation, so care should be taken to avoid contact when using it. Avoid inhaling dust and vapors and ventilate properly. Storage should be placed in a dry, cool place, away from fire and oxidant.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: 5-HYDROXY-2-METHOXYPHENYLBORONIC ACID Request for quotation
CAS: 1072952-43-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1072952-43-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 5-HYDROXY-2-METHOXYPHENYLBORONIC ACID Request for quotation
CAS: 1072952-43-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1072952-43-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
View History
